COVID-19 Research
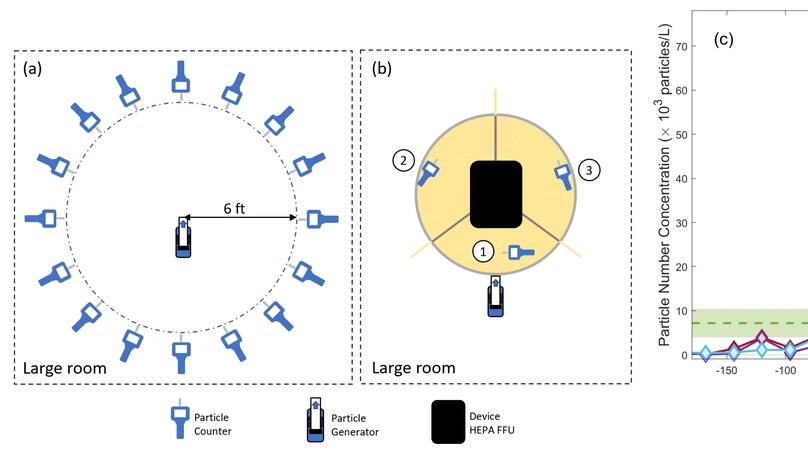
Here we present a device that suppresses transfer of aerosol between nearby seating areas through the use of optically transparent, sound transmitting barriers and HEPA fan filter unit (FFU). A potential application of this device is to lower the risk of respiratory disease transmission in face-to-face, maskless meetings between individuals in a university setting. We evaluate overall aerosol transmission between users of the device. This is done for two different physical settings: a large space, such as a library, and a small space, such as an enclosed study room. We find that the device can provide lower aerosol transmission compared to the typical transmission between two individuals wearing surgical face masks separated by six feet.
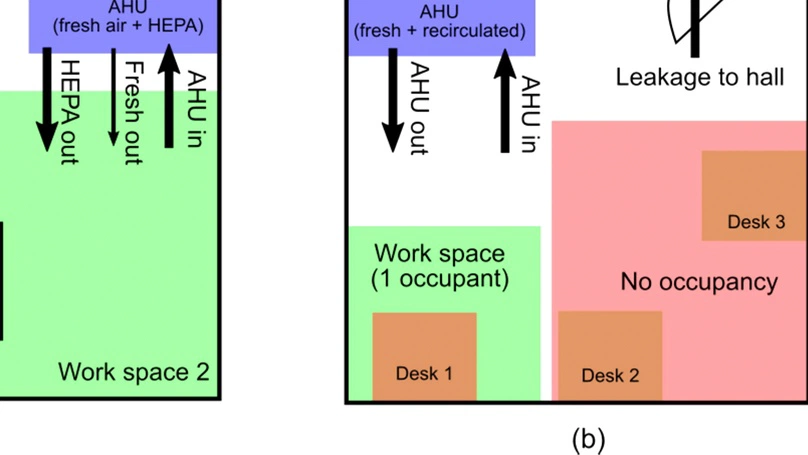
Bioaerosols are known to be an important transmission pathway for SARS-CoV-2. We report a framework for estimating the risk of transmitting SARS-CoV-2 via aerosols in laboratory and office settings, based on an exponential dose-response model and analysis of air flow and purification in typical heating, ventilation, and air conditioning (HVAC) systems. High-circulation HVAC systems with high-efficiency particulate air (HEPA) filtration dramatically reduce exposure to the virus in indoor settings, and surgical masks or N95 respirators further reduce exposure. As an example of our risk assessment model, we consider the precautions needed for a typical experimental physical science group to maintain a low risk of transmission over six months of operation. We recommend that, for environments where fewer than five individuals significantly overlap, work spaces should remain vacant for between one (high-circulation HVAC with HEPA filtration) to six (low-circulation HVAC with no filtration) air exchange times before a new worker enters in order to maintain no more than 1% chance of infection over six months of operation in the workplace. Our model is readily applied to similar settings that are not explicitly given here. We also provide a framework for evaluating infection mitigation through ventilation in multiple occupancy spaces.